Epigenetics
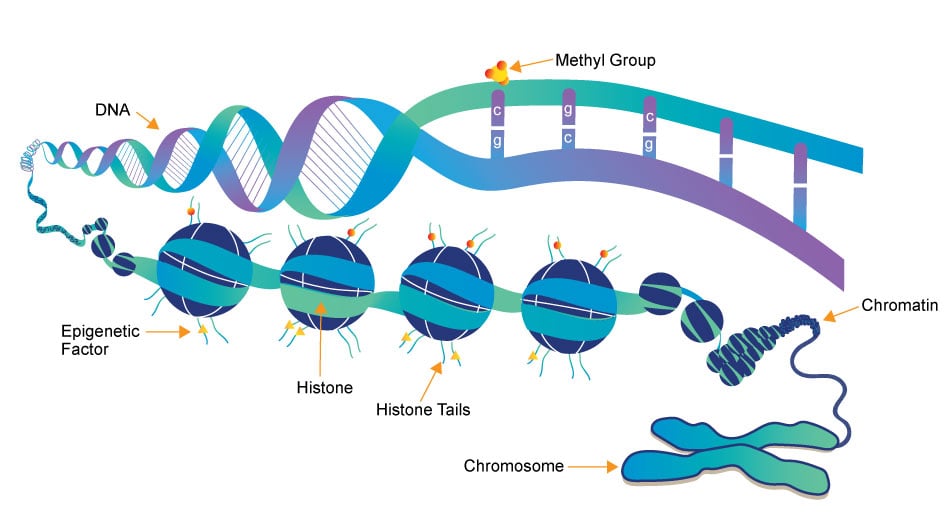

Epigenetics research delves into the molecular mechanisms that control gene expression and cellular traits without altering the underlying DNA sequence. One crucial aspect of this field is the role of small molecules, which act as powerful regulators of epigenetic modifications. These small compounds, typically comprising a few dozen to a few hundred atoms, have emerged as essential tools in understanding and manipulating the epigenome.
- DNA Methylation Inhibitors: Small molecules like 5-azacytidine and 5-aza-2'-deoxycytidine are DNA methyltransferase inhibitors. They block the addition of methyl groups to DNA, leading to DNA demethylation. This can reactivate silenced genes, potentially offering therapeutic avenues for conditions like cancer.
- HDAC inhibitors: HDACs remove acetyl groups from histone proteins, contributing to gene repression. Small molecule HDAC inhibitors, such as Vorinostat and Romidepsin, can reverse this process by increasing histone acetylation, allowing genes to be more accessible for transcription. These inhibitors are being explored for cancer therapy and other conditions.
- Histone Methyltransferase Inhibitors: Small molecules like GSK126 inhibit specific histone methyltransferases, affecting histone methylation patterns. This can alter gene expression, making them promising candidates for cancer and other diseases with epigenetic dysregulation.
- RNA Modulators: Small molecules can also target non-coding RNAs involved in epigenetic regulation. For instance, small molecules called small interfering RNAs (siRNAs) can be designed to target and degrade specific long non-coding RNAs, influencing gene expression.
- Epigenetic Reader Domain Inhibitors: These small molecules target proteins that recognize and bind to specific epigenetic marks. Examples include inhibitors of bromodomain-containing proteins (BET inhibitors), which can disrupt gene regulation by interfering with protein-DNA interactions.
Small molecules in epigenetics research not only provide insights into the fundamental biology of gene regulation but also hold immense promise for developing novel therapeutics. Their ability to selectively modulate specific epigenetic marks and pathways has led to ongoing clinical trials and drug development efforts for various diseases, including cancer, neurological disorders, and inflammatory conditions. Understanding and harnessing the power of these small molecules is at the forefront of modern epigenetics research, offering new hope for precision medicine and targeted therapies.
3 key components involved in the regulation of epigenetic modifications
Epigenetics Writer
Epigenetics writers are enzymes responsible for adding chemical marks or modifications to DNA or histone proteins. These marks include DNA methylation (addition of methyl groups to DNA) and histone modifications (such as acetylation, methylation, phosphorylation, etc.).
Epigenetics Reader
Function: Epigenetics readers are proteins that can recognize and bind to specific epigenetic marks on DNA or histones. These reader proteins interpret the epigenetic code and facilitate downstream cellular processes, such as gene activation or repression.
Epigenetics Eraser
Function: Epigenetics erasers are enzymes responsible for removing or reversing epigenetic marks on DNA or histones. This process allows for the dynamic regulation of gene expression and the resetting of epigenetic states during various stages of development and in response to environmental changes.
- Trifloxystrobin (CGA 279202) is a fungicide, with EC50s of 23.0 μg/L and 1.7 μg/L for Daphnia magna neonate and embryos, respectively, after treatment for 48 h.
-
anticancer agent
5,7,4'-Trimethoxyflavone is isolated from Kaempferia parviflora (KP) that is a famous medicinal plant from Thailand. 5,7,4'-Trimethoxyflavone induces apoptosis, as evidenced by increments of sub-G1 phase, DNA fragmentation, annexin-V/PI staining, the Bax/Bcl-xL ratio, proteolytic activation of caspase-3, and degradation of poly (ADP-ribose) polymerase (PARP) protein.5,7,4'-Trimethoxyflavone is significantly effective at inhibiting proliferation of SNU-16 human gastric cancer cells in a concentration dependent manner. -
Caspase-1/4 inhibitor
VX-765 is a novel Caspase-1 inhibitor,which is an enzyme that controls the generation of two cytokines, IL-1b and IL-18.- Joseph Flores, .et al. , Cell Death Dis, 2022, Oct 11;13(10):864 PMID: 36220815
- Arjun Thapa, .et al. , Leukemia, 2021, 23 February
- Prenitha Mercy Ignatius Arokia Doss, .et al. , Cell Rep, 2021, Mar 9;34(10):108833 PMID: 33691111
- Joseph Flores, .et al. , Nat Commun, 2020, Sep 11;11(1):4571 PMID: 32917871
- Joseph Flores, .et al. , Nat Commun, 2018, 9: 3916 PMID: 30254377
- Mohamed F. Ali, .et al. , Front Immunol, 2017, 8: 1504 PMID: 29170665
- García-Fernández A, .et al. , J Control Release, 2017, Feb 28;248:60-70 PMID: 28069553
- Jiujiu Yu, .et al. , Proc Natl Acad Sci U S A, 2014, 111(43): 15514-15519 PMID: 25313054
-
Caspase activator
PAC-1 is an activator of procaspase-3 induces apoptosis in cancer cells with EC50 of 2.08 μM.- Hirosumi Tamura, .et al. , Oncol Rep, 2018, Aug; 40(2): 635-646 PMID: 29917168
-
Apoptosis Activator
Apoptosis Activator 2 strongly induces caspase-3 activation, PARP cleavage, and DNA fragmentation which leads to the destruction of cells (Apaf-1 dependent) with IC50 of ~4 μM, inactive to HMEC, PREC, or MCF-10A cells -
Pan Caspase Inhibitor
Z-VAD-FMK is a cell-permeable, irreversible pan-caspase inhibitor. Inhibits caspase processing and apoptosis induction in tumor cells in vitro (IC50 = 0.0015 - 5.8 mM). Active in vivo.- Chantal-Kristin Wenzel, .et al. , Toxicol In Vitro, 2023, Sep;91:105625 PMID: 37268255
- Joshua H Choe, .et al. , Cancer Discov, 2023, May 4;13(5):1250-1273 PMID: 37067901
- Yu-Chih Hsu, .et al. , Int J Med Sci, 2023, 20(4): 444-454 PMID: 37057216
- Debin Qi, .et al. , Cell Cycle, 2023, Apr;22(7):818-828 PMID: 36482709
- Ming-Chun Hung, .et al. , Cell Biosci, 2022, 12: 122 PMID: 35918763
- Kaori Kanemaru, .et al. , Nat Commun, 2022, May 9;13(1):2347 PMID: 35534464
- Chie Ishikawa, .et al. , Invest New Drugs, 2022, Aug;40(4):718-727 PMID: 35477814
- Aida Falgas, .et al. , Biomed Pharmacother, 2022, Jun;150:112940 PMID: 35421785
- Sumire Suzuki, .et al. , Oncol Rep, 2022, Feb;47(2):40 PMID: 34958115
- Lina Y Abou Zeid, .et al. , Cell Stress Chaperones, 2022, Jan;27(1):11-25 PMID: 34719748
- Simon Boudreault, .et al. , Viruses, 2022, Dec 3;14(12):2710 PMID: 36560714
- Kosar Jabbari, .et al. , Cancers (Basel), 2021, Sep 8;13(18):4521 PMID: 34572749
- Xiaoyue Deng, .et al. , Invest Ophthalmol Vis Sci, 2021, May 3;62(6):1 PMID: 33938913
- Liqun Zhao, .et al. , Apoptosis, 2021, Apr 27 PMID: 33905036
- Sofia Giacosa, .et al. , Cancers (Basel), 2021, Feb 2;13(3):576 PMID: 33540838
- Hannes Schmid, .et al. , Haematologica, 2021, Jan 14 PMID: 33440919
- Hua Xu, .et al. , Cancers (Basel), 2020, Apr; 12(4): 831 PMID: 32235588
- El-Ashmawy NE, .et al. , Pathol Oncol Res, 2020, Jan 4 PMID: 31902118
- Schneider D, .et al. , Clin Epigenetics, 2019, Jul 2;11(1):98 PMID: 31266541
- Fujiki K, .et al. , Cell Death Differ, 2019, Feb 25 PMID: 30804470
- T Yamamoto, .et al. , Cell Microbiol, 2019, 2019 PMID: 30702185
- Maeda N, .et al. , J Biol Chem, 2019, Nov 13. pii: jbc.RA119.010379 PMID: 31723031
- Miyazawa S, .et al. , Breast Cancer, 2019, Oct 17 PMID: 31625014
- Yeh HT, .et al. , Eur J Pharmacol, 2019, Sep 10;863:172658 PMID: 31518562
- D. Bosc, .et al. , Sci Rep, 2018, 8: 11653 PMID: 30076329
- Tsukamoto H, .et al. , J Biol Chem, 2018, Jun 29;293(26):10186-10201 PMID: 29760187
- Hiroshi Kuribayashi, .et al. , Cell Death Dis, 2018, Sep; 9(9): 891 PMID: 30166529
- Matsuo J, .et al. , Can J Microbiol, 2018, Oct 18:1-9 PMID: 30336068
- Orly Ravid, .et al. , Front Cell Neurosci, 2018, 12: 359 PMID: 30459557
- Im E, .et al. , Life Sci, 2018, Sep 15;209:259-266 PMID: 30107166
- Szalai P, .et al. , Cell Calcium, 2018, Dec;76:48-61 PMID: 30261424
- Im E, .et al. , Life Sci, 2018, Jan 1;192:286-292 PMID: 29128513
- Iriyama N, .et al. , Leuk Lymphoma, 2018, Jun;59(6):1439-1450 PMID: 28918692
- Birte Plitzko, .et al. , J Biol Chem, 2017, Dec 22; 292(51): 21102-21116 PMID: 29074620
- Akashi E, .et al. , Oncol Rep, 2017, Jul;38(1):506-514 PMID: 28586026
-
Caspase-6 inhibitor
Z-VEID-FMK is the specific recognition sequence for caspase-6/Mch2. Z-VEID-FMK is a synthetic peptide that irreversibly inhibits activity of VEID-dependent caspases (e.g., caspase-6). The inhibitor is designed as a methyl ester to facilitate cell permeability. -
Caspase-2 inhibitor
Z-VDVAD-FMK is a cell-permeable, irreversible inhibitor of caspase-2. Caspase inhibitors play an important role in investigating biological processes. -
Caspase-3 inhibitor
Z-DQMD-FMK is a synthetic peptide that irreversibly inhibits the activity of Caspase-3.- Jason Segura, .et al. , PLoS One, 2023, Feb 13;18(2):e0281087 PMID: 36780482
-
caspase 8 inhibitor
Z-IETD-FMK is a specific inhibitor of caspase 8.- Jason Segura, .et al. , PLoS One, 2023, Feb 13;18(2):e0281087 PMID: 36780482
- Liqun Zhao, .et al. , Apoptosis, 2021, Apr 27 PMID: 33905036
-
Apoptosis inducer
LY573636 is a potent anti-tumor agent, which causes growth arrest and apoptosis of a variety of human solid tumors in vitro and in vivo. LY573636-induced apoptosis occurs by a mitochondrial-targeted mechanism.- Ting TC, .et al. , Cell Rep, 2019, Nov 5;29(6):1499-1510.e6 PMID: 31693891
- Ting Han, .et al. , Science, 2017, Apr 28;356(6336). pii: eaal3755 PMID: 28302793
-
Caspase-3 Inhibitor
Z-DEVD-FMK is a specific, irreversible Caspase-3 inhibitor.- Roi Ankawa, .et al. , Dev Cell, 2021, Jul 12;56(13):1900-1916 PMID: 34197726
- Yosefzon Y, .et al. , Mol Cell, 2018, May 17;70(4):573-587.e4 PMID: 29775577
- Akashi E, .et al. , Oncol Rep, 2017, Jul;38(1):506-514 PMID: 28586026
- Wenquan Liang, .et al. , Sci Rep, 2016, 6: 38267 PMID: 27905569
- 25-hydroxy Cholesterol is a side-chain substituted oxysterol derived from dietary cholesterol that inhibits the cleavage of sterol regulatory element binding proteins (SREBPs) to suppress endogenous cholesterol synthesis in various cell types
-
Caspase inhibitor
Nivocasan is a novel caspase-inhibitor has demonstrated hepatoprotective activity in fibrosis/apoptosis animal models. -
Caspase-9 Inhibitor
Z-LEHD-FMK is a synthetic peptide that irreversibly inhibits caspase-9 and related caspase activity.- Jason Segura, .et al. , PLoS One, 2023, Feb 13;18(2):e0281087 PMID: 36780482
- Wenquan Liang, .et al. , Sci Rep, 2016, 6: 38267 PMID: 27905569
-
osteoblast differentiation inducer
Asperosaponin VI, A saponin component from Dipsacus asper wall, induces osteoblast differentiation through BMP?\2/p38 and ERK1/2 pathway. Asperosaponin ?? inhibits apoptosis in hypoxia-induced cardiomyocyte by increasing the Bcl-2/Bax ratio and decreasing active caspase-3 expression, as well as enhancing of p-Akt and p-CREB. -
Caspase inhibitor
Q-VD-OPH is a selective, brain and cell permeable, highly potent and irreversible inhibitor of caspase-3 ( IC50=25nm), caspase-1 (IC50=50nM), caspase-8 (IC50=100nM) and caspase-9 (IC50=430nM).- Anna L Koessinger, .et al. , Cell Death Differ, 2022, Apr 26 PMID: 35473984
- Florian J Bock, .et al. , Nat Commun, 2021, Nov 12;12(1):6572 PMID: 34772930
- Davide Pradella, .et al. , Nat Commun, 2021, 12: 4872 PMID: 34381052
- Haschka MD, .et al. , Cell Death Differ, 2020, Feb 3 PMID: 32015503
-
Caspase inhibitor
Z-WEHD-FMK is a synthetic peptide that irreversibly inhibits caspase-5 and related caspase activity. -
Caspase-3/7 Inhibitor
Caspase-3/7 Inhibitor I is a potent, cell-permeable, and specific, reversible inhibitor of caspase-3 (Ki = 60 nM) and caspase-7 (Ki = 170 nM). -
Fluorogenic caspase substrate
Ac-LEHD-AFC, fluorogenic caspase substrate. Analog of the caspase-9 substrate, LEHD-AFC. -
Caspase-3 inhibitor
AZ 10417808, selective non-peptide inhibitor of caspase-3 (Ki = 247 nM); displays > 40-fold selectivity over caspases 1, 2, 6, 7 and 8 (Ki > 10 uM). -
Caspase-3 inhibitor
Ivachtin is a potent, cell-permeable, reversible, non-competitive inhibitor of Caspase-3 (IC50 = 23 nM).- Yosefzon Y, .et al. , Mol Cell, 2018, May 17;70(4):573-587.e4 PMID: 29775577
-
Fluorogenic caspase substrate
Ac-LEHD-AFC is a fluorogenic substrate for caspase-4, caspase-5, and caspase-9. -
Caspase Inhibitor
Ac-DEVD-CHO is an aldehyde peptide and a CPP32/Apopain Inhibitor. -
Caspase-1 inhibitor
Z-YVAD-FMK is a potent cell-permeable and irreversible inhibitor of caspase-1.- Yuping Fan, .et al. , Sci Transl Med, 2021, Jul 28;13(604):eabb1069 PMID: 34321317
-
Caspase inhibitor
Z-VAD(OH)-FMK is an irreversible tripeptide inhibitor of all caspases. - Thevetiaflavone could upregulate the expression of Bcl?2 and downregulate that of Bax and caspase?3.
-
insecticidal and anticancer
Destruxin B, isolated from entomopathogenic fungus Metarhizium anisopliae, is one of the cyclodepsipeptides with insecticidal and anticancer activities. Destruxin B induces apoptosis via a Bcl-2 Family-dependent mitochondrial pathway in human nonsmall cell lung cancer cells. - Lycopodine, a pharmacologically important bioactive component derived from Lycopodium clavatumspores, triggers apoptosis by modulating 5-lipoxygenase, and depolarizing mitochondrial membrane potential in refractory prostate cancer cells without modulating p53 activity. Lycopodine inhibits proliferation of HeLa cells through induction of apoptosis via caspase-3 activation.
- Guggulsterone is a plant sterol derived from the gum resin of the tree Commiphora wightii. Guggulsterone inhibits the growth of a wide variety of tumor cells and induces apoptosis through down regulation of antiapoptotic gene products (IAP1, xIAP, Bfl-1/A1, Bcl-2, cFLIP and survivin), modulation of cell cycle proteins (cyclin D1 and c-Myc), activation of caspases and JNK, inhibition of Akt.
- Phenoxodiol, a synthetic analog of Genestein, activates the mitochondrial caspase system, inhibits XIAP (an apoptosis inhibitor), and sensitizes the cancer cells to Fas-mediated apoptosis.
-
Caspase-1 substrate
Ac-YVAD-pNA is a specific Caspase-1 substrate. Ac-YVAD-pNA can be used to detect Caspase-1 activity. Caspase-1 is a key mediator of inflammatory processes.









