Epigenetics
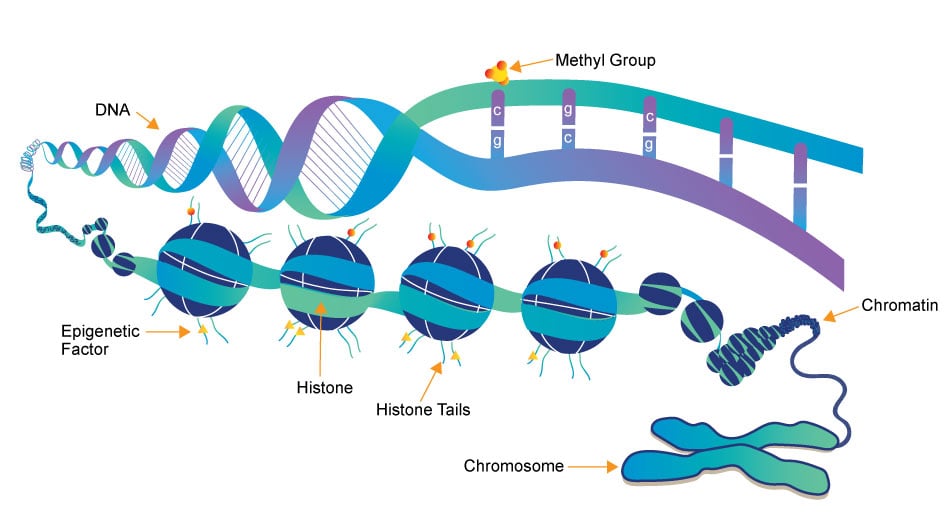

Epigenetics research delves into the molecular mechanisms that control gene expression and cellular traits without altering the underlying DNA sequence. One crucial aspect of this field is the role of small molecules, which act as powerful regulators of epigenetic modifications. These small compounds, typically comprising a few dozen to a few hundred atoms, have emerged as essential tools in understanding and manipulating the epigenome.
- DNA Methylation Inhibitors: Small molecules like 5-azacytidine and 5-aza-2'-deoxycytidine are DNA methyltransferase inhibitors. They block the addition of methyl groups to DNA, leading to DNA demethylation. This can reactivate silenced genes, potentially offering therapeutic avenues for conditions like cancer.
- HDAC inhibitors: HDACs remove acetyl groups from histone proteins, contributing to gene repression. Small molecule HDAC inhibitors, such as Vorinostat and Romidepsin, can reverse this process by increasing histone acetylation, allowing genes to be more accessible for transcription. These inhibitors are being explored for cancer therapy and other conditions.
- Histone Methyltransferase Inhibitors: Small molecules like GSK126 inhibit specific histone methyltransferases, affecting histone methylation patterns. This can alter gene expression, making them promising candidates for cancer and other diseases with epigenetic dysregulation.
- RNA Modulators: Small molecules can also target non-coding RNAs involved in epigenetic regulation. For instance, small molecules called small interfering RNAs (siRNAs) can be designed to target and degrade specific long non-coding RNAs, influencing gene expression.
- Epigenetic Reader Domain Inhibitors: These small molecules target proteins that recognize and bind to specific epigenetic marks. Examples include inhibitors of bromodomain-containing proteins (BET inhibitors), which can disrupt gene regulation by interfering with protein-DNA interactions.
Small molecules in epigenetics research not only provide insights into the fundamental biology of gene regulation but also hold immense promise for developing novel therapeutics. Their ability to selectively modulate specific epigenetic marks and pathways has led to ongoing clinical trials and drug development efforts for various diseases, including cancer, neurological disorders, and inflammatory conditions. Understanding and harnessing the power of these small molecules is at the forefront of modern epigenetics research, offering new hope for precision medicine and targeted therapies.
3 key components involved in the regulation of epigenetic modifications
Epigenetics Writer
Epigenetics writers are enzymes responsible for adding chemical marks or modifications to DNA or histone proteins. These marks include DNA methylation (addition of methyl groups to DNA) and histone modifications (such as acetylation, methylation, phosphorylation, etc.).
Epigenetics Reader
Function: Epigenetics readers are proteins that can recognize and bind to specific epigenetic marks on DNA or histones. These reader proteins interpret the epigenetic code and facilitate downstream cellular processes, such as gene activation or repression.
Epigenetics Eraser
Function: Epigenetics erasers are enzymes responsible for removing or reversing epigenetic marks on DNA or histones. This process allows for the dynamic regulation of gene expression and the resetting of epigenetic states during various stages of development and in response to environmental changes.
-
CDK inhibitor
Alsterpaullone (9-Nitropaullone;NSC 705701) is a potent CDK inhibitor, with IC50s of 35 nM, 15 nM, 200 nM and 40 nM for CDK1/cyclin B, CDK2/cyclin A, CDK2/cyclin E and CDK5/p35, respectively. -
CDC7 inhibitor
Simurosertib (TAK-931) is a selective cycle 7 (CDC7) kinase inhibitor, with an IC50<0.3 nM. -
CDK8 inhibitor
CCT-251921 is a potent, selective, and orally bioavailable CDK8 inhibitor with an IC50 of 2.3 nM. -
CDK inhibitor
Voruciclib hydrochloride is a clinical stage orally active and selective CDK inhibitor with Ki values of 0.626 nM-9.1 nM. -
dual Cdc7/Cdk9 inhibitor
PHA-767491 hydrochloride is a dual Cdc7/Cdk9 inhibitor, with IC50s of 10 nM and 34 nM, respectively. -
CDK9 inhibitor
CDKI-73 is a potent CDK9 inhibitor with Ki of 4 nM; shows selective toxicity to CLL cells(LD50=80 nM) versus normal B cell and normal CD34+ cell(LD50>20 uM). -
CDK8 inhibitor
SEL120-34A HCl is a potent, selective, orally available, ATP-competitive CDK8 inhibitor, with IC50s of 4.4 nM and 10.4 nM for CDK8/CycC and CDK19/CycC, respectively, with antitumor activity. -
PTEF/CDK9 inhibitor
(±)-BAY-1251152 is a racemic mixture of BAY-1251152. BAY-1251152 is a potent and highly selective PTEF/CDK9 inhibitor. -
PTEF/CDK9 inhibitor
(-)-BAY-1251152 is an enanthiomer of BAY-1251152 with rotation (-). BAY-1251152 is a potent and highly selective PTEF/CDK9 inhibitor. -
CDK4/CDK6 inhibitor
Lerociclib dihydrochloride (G1T38 dihydrochloride) is a potent and selective inhibitor of CDK4/CDK6, with IC50s of 1 nM and 2 nM for CDK4/CyclinD1 and CDK6/CyclinD3, respectively. -
CDK7 inhibitor
BS-181 hydrochloride is a highly selective CDK7 inhibitor with IC50 of 21 nM, and > 40-fold selective for CDK7 than CDK1, 2, 4, 5, 6, or 9. -
PKC inhibitor
Bisindolylmaleimide X hydrochloride (BIM-X hydrochloride) is a potent and selective protein kinase C (PKC) inhibitor. -
CDK4/6 inhibitor
Trilaciclib hydrochloride is a CDK4/6 inhibitor with IC50s of 1 nM and 4 nM for CDK4 and CDK6, respectively. -
ATP-competitive CDK2 and CDK5 inhibitor
PNU112455A hydrochloride is an ATP-competitive CDK2 and CDK5 inhibitor. -
CDK4/6 inhibitor
Ribociclib hydrochloride (LEE011 hydrochloride) is a highly specific CDK4/6 inhibitor with IC50 values of 10 nM and 39 nM, respectively,- Vijaya Bharti, .et al. , Cell Rep, 2022, Dec 20;41(12):111826 PMID: 36543138
-
CDK6 degrader
BSJ-03-123 is a potent and novel CDK6-selective small-molecule degrader (PROTAC). -
dual inhibitor of protein kinase and CDK
6-(Dimethylamino)purine is a dual inhibitor of protein kinase and CDK.








