-
IDH1 inhibitor
AGI-5198, also know as IDH-C35, is the a very potent and selective mutant IDH1 inhibitor that was shown to potential anticancer activity. -
IDH1 inhibitor
AG-120 is an orally available inhibitor of isocitrate dehydrogenase type 1 (IDH1), with potential antineoplastic activity. -
IDH2 inhibitor
Enasidenib is a potent and selective IDH2 inhibitor with potential anticancer activity (IDH2 = Isocitrate dehydrogenase 2).- Mohammed NadimSardoiwala, .et al. , Materials Science and Engineering: C, 2021, Dec 24
-
IDH1 Inhibitor
Vorasidenib, also known as AG-881, is a potent and selective orally available inhibitor of mutated forms of both isocitrate dehydrogenase type 1 (IDH1, IDH1 [NADP+] soluble) in the cytoplasm and type 2 (IDH2, isocitrate dehydrogenase [NADP+], mitochondrial) in the mitochondria, with potential antineoplastic activity. -
mIDH1 inhibitor
BAY-1436032 is a potent, selective and orally available inhibitor of mutant Isocitrate Dehydrogenase 1 (mIDH1). -
IDH1 inhibitor
DH1 Inhibitor 2 is a potent IDH1 inhibitor via a direct covalent modification of His315, with an IC50 of 110 nM. -
IDH1 inhibitor
Olutasidenib is a potent, selective inhibitor of mutant Isocitrate dehydrogenase (IDH)1 for the treatment of acute myeloid leukemia. -
IDH1 inhibitor
Mutant IDH1-IN-4 (compound 434) is an inhibitor of mutant Isocitrate dehydrogenase 1 (IDH 1), with IC50 values of ?? 0.5 μM for mutant IDH1 in R132H, HT1080 and U87R132H cells. -
IDH1 inhibitor
IDH1 Inhibitor 3 (compound 6f) is a mutant isocitric dehydrogenase 1 (IDH1) inhibitor, with an IC50 of 45 nM for IDH1R132H. -
IDH1 inhibitor
IDH1 Inhibitor 1 is a potent, orally bioavailable, brain-penetrant and selective mutant IDH1 inhibitor with IC50s of 0.021 μM, 0.045 μM, and 2.52 μM for IDH1R132H, IDH1R132C, and IDH1WT, respectively. Anticancer activity. -
mutant IDH1 R132H inhibitor
Mutant IDH1 inhibitor is a potent mutant IDH1 R132H inhibitor with IC50 of < 72 nM. -
IDH inhibitor
Mutant IDH1-IN-2 is a inhibitor of mutant Isocitrate dehydrogenase (IDH) proteins, with IC50 of in LS-MS biochemical assay, IC50 of 16.6 nM in Fluorescence biochemical assay. -
mutant-selective IDH1 inhibitor
Mutant IDH1-IN-1 is a mutant-selective IDH1 inhibitor with with IC50s of 4, 42, 80 and 143 nM against mutant IDH1 R132C/R132C, IDH1 R132H/R132H, IDH1 R132H/WT and wild type IDH1, respectively.
Endocrinology-Hormones
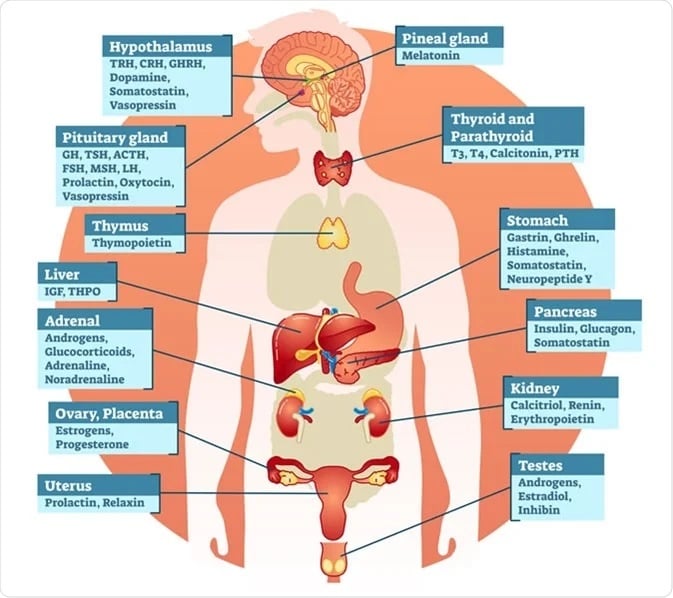

Small molecules play a pivotal role in Endocrinology Research. These are low molecular weight compounds that have a significant impact on the endocrine system, hormones, and their receptors. Here are some key aspects of how small molecules are involved in this field:
- Hormone Mimetics and Inhibitors: Small molecules are used to develop synthetic compounds that mimic the actions of hormones or inhibit their effects. For example, drugs like metformin for diabetes management and selective estrogen receptor modulators (SERMs) for breast cancer treatment are used to either mimic or block hormonal activity.
- Receptor Modulation: Small molecules can bind to hormone receptors and modulate their activity. This is crucial in developing drugs that target specific hormone receptors, like the use of small molecule agonists and antagonists to regulate thyroid hormone receptors.
- Metabolism Regulation: Endocrinology research often focuses on metabolism and how hormones like insulin regulate it. Small molecules are employed to understand and develop drugs targeting enzymes involved in metabolism, such as glucagon-like peptide-1 (GLP-1) agonists for diabetes treatment.
- Steroid Hormone Production: Small molecules may be utilized to influence the production of steroid hormones in the adrenal glands or gonads. This is essential for conditions like Cushing's syndrome or polycystic ovary syndrome (PCOS).
- Hormone Assays: In laboratory research, small molecules are used as tracers or markers in hormone assays. For instance, small molecule fluorophores can be attached to antibodies to detect hormone levels in blood samples.
Drug Development: Endocrinology research relies on small molecules as potential drug candidates. Researchers design and test small molecules for their effectiveness in modulating hormonal pathways, with the goal of developing new therapies for endocrine disorders.
In summary, small molecules are indispensable tools in Endocrinology Research, enabling scientists to better understand the endocrine system's intricacies and develop novel treatments for a wide range of hormonal disorders and conditions. Their versatility and specificity make them valuable assets in advancing our knowledge of endocrinology and improving patient care.
Endocrinology Disease Products
Endocrinology Research Products
Kisspeptin Receptor
Leptin Receptors
Melanocortin (MC) Receptors
Mineralocorticoid Receptors
Galanin Receptors
TRH Receptors
Ghrelin Receptors
Natriuretic Peptide Receptors
NPY Receptors
Motilin Receptor
PTH Receptor








